Electrolytic Polishing
1.Definition of electrolytic polishing
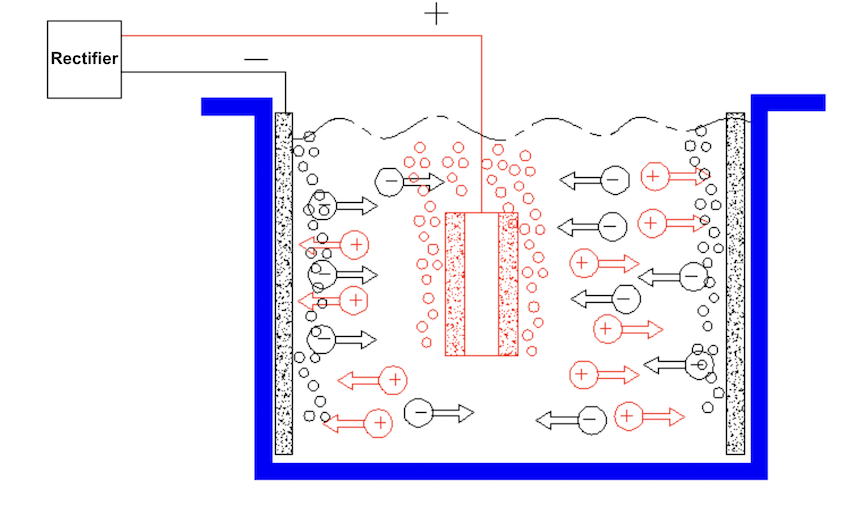
Electrolytic polishing (Electrolytic polishing) is also known as electrochemical polishing. It takes the polished workpiece as the anode and the insoluble metal as the cathode.
The two poles are immersed in the electrolytic cell at the same time, and direct current is used to produce selective anode dissolution, thus increasing the brightness of the workpiece surface.

2.Principle of electrolytic polishing
The principle of electrolytic polishing is mainly based on the mucosal theory. The theory is mainly as follows: the metal ions separated from the workpiece and the phosphoric acid in the polishing liquid form a phosphate film adsorbed on the workpiece surface. This kind of mucous membrane is thinner at the bulge and thicker at the concave. Due to the high current density at the bulge, the mucous membrane dissolves quickly.
3.Electrolytic polishing characteristics
(1) The internal and external color is consistent, luster lasting, mechanical polishing can not throw to the concave can also be leveled.
(2) High production efficiency and low cost.
(3) Increase the corrosion resistance of the workpiece surface.
4. It can remove burrs on the surface of parts
5. Electrolytic workpiece surface is not easy to litter and breed bacteria
6. Environmental protection universal electrolyte characteristics
A, polishing liquid does not contain chromium ions, in line with today's environmental protection requirements, saving environmental protection equipment investment and low cost of waste water treatment; It can be authenticated by ROSH.
B, polishing current density is small, low electricity, electric energy consumption is more than 1 times lower than the traditional process;
C, high polishing efficiency, good quality, within a few minutes can be thrown to the mirror bright effect;
D, polishing liquid long service life, easy to maintain and manage.
If you have another question please feel free to contact email: enquiry@abcrapid.com.
Search
Recent Post













